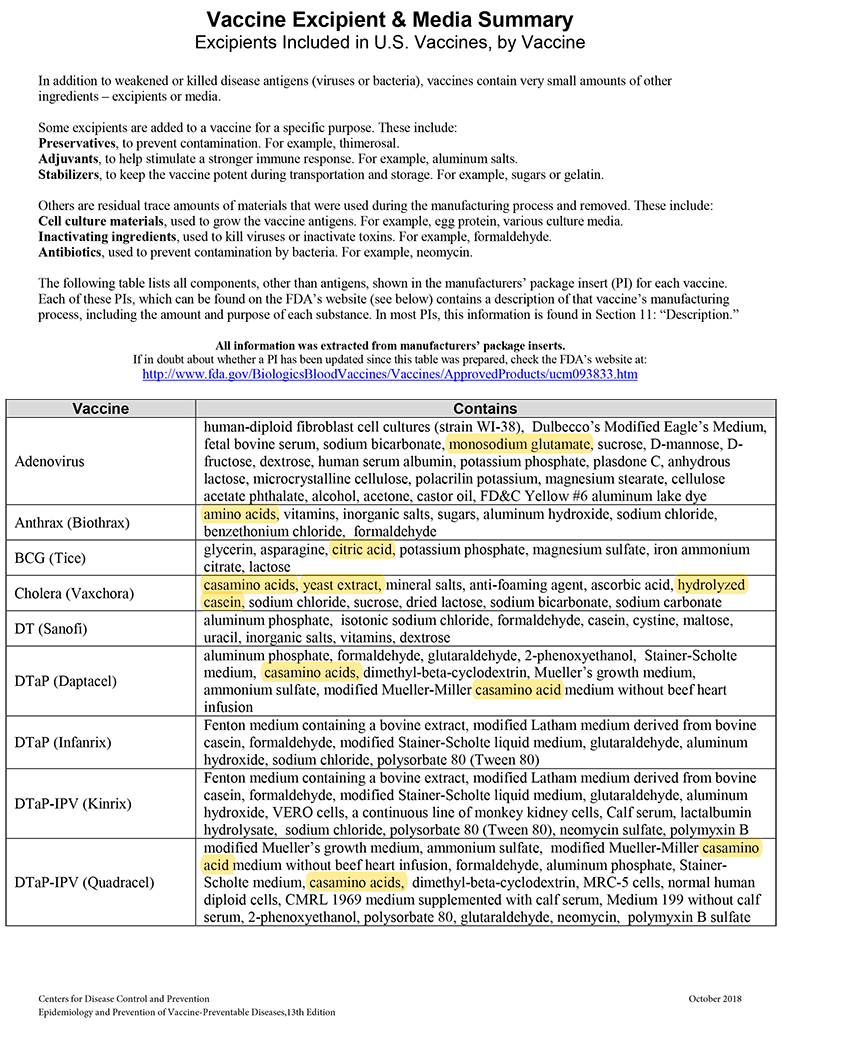
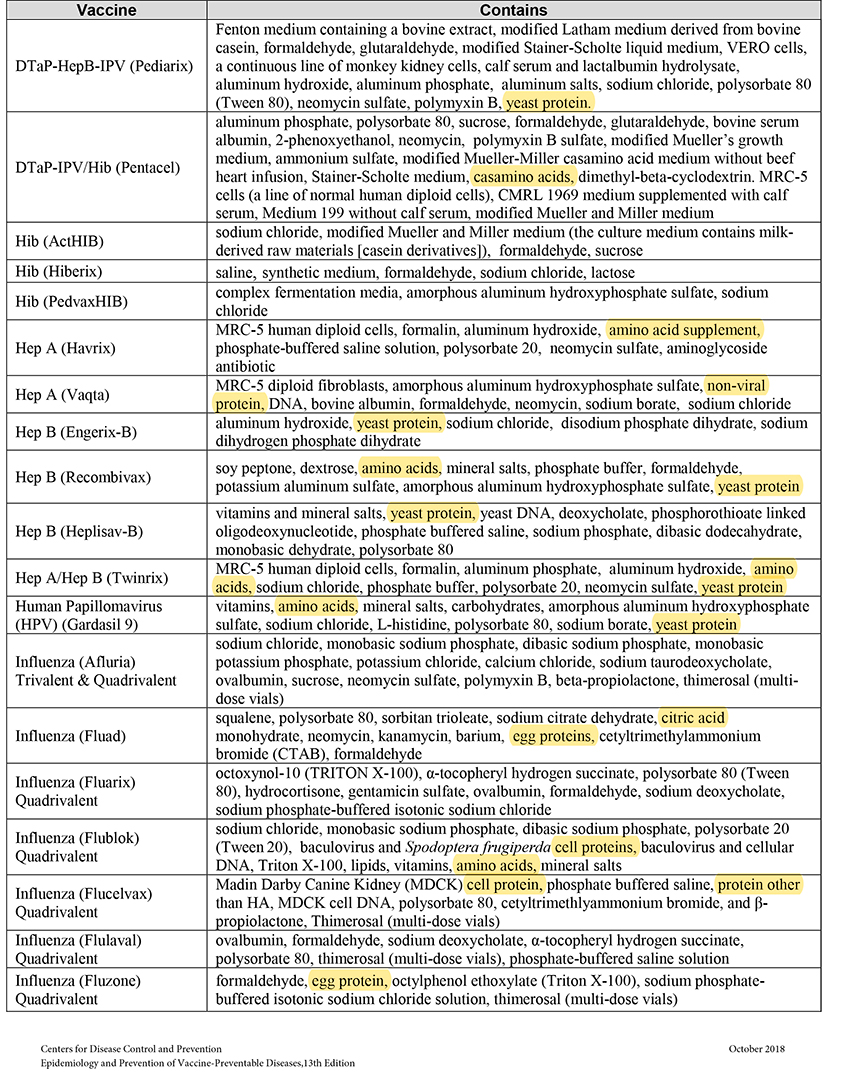
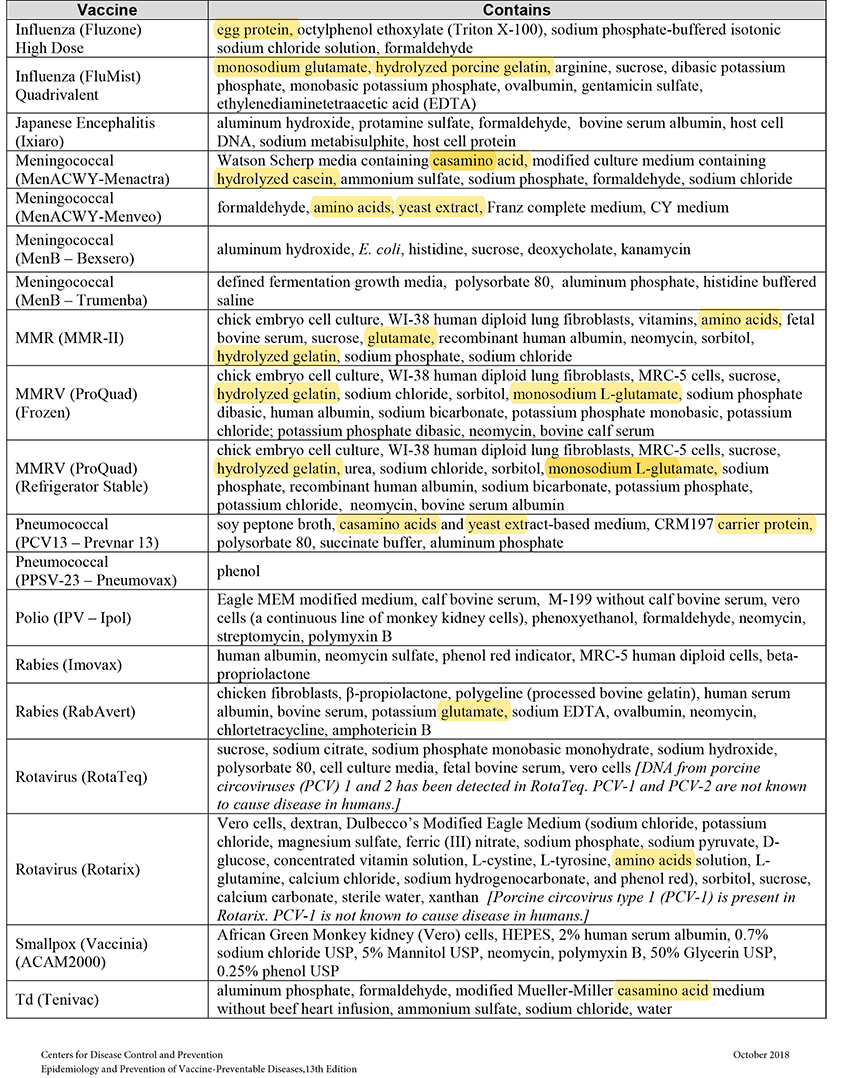
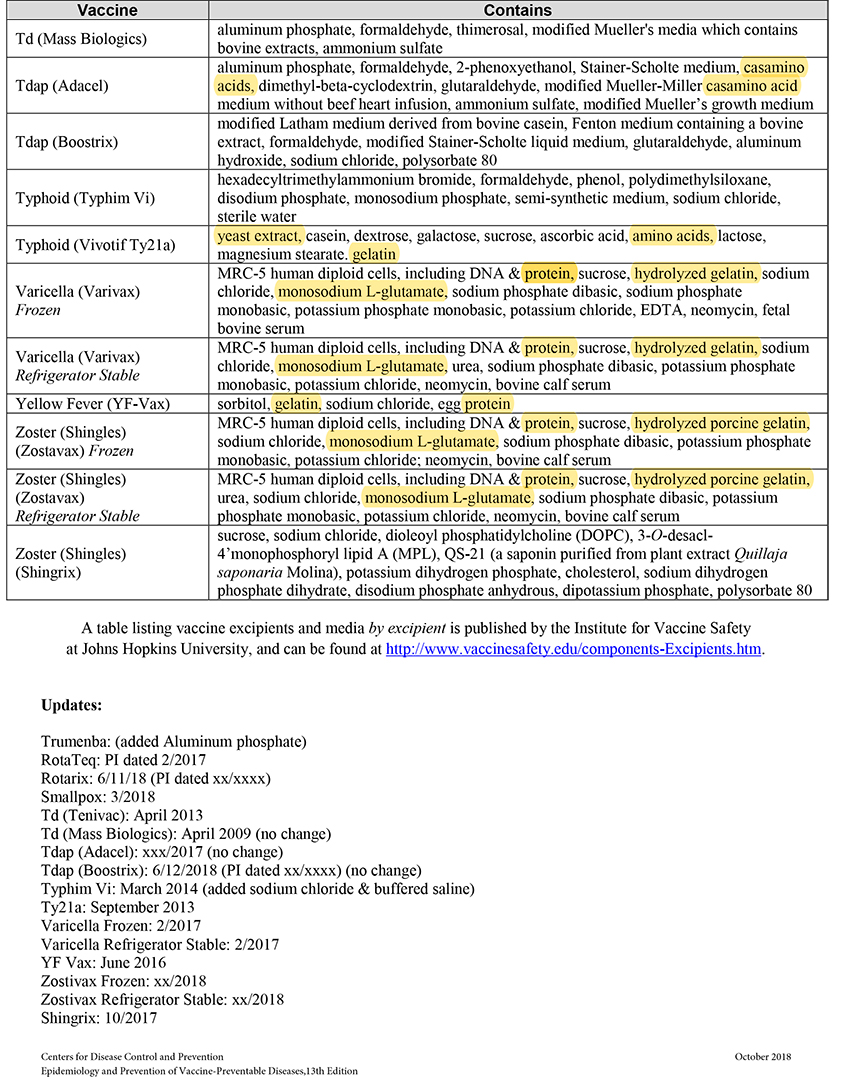
When MSG and other sources of toxic manufactured free glutamic acid (MfG) are used in pharmaceuticals as excipients (a.k.a. “inactive” ingredients), manufacturers are not required to share that information with the public. This past June the Truth in Labeling Campaign reviewed the excipients found in vaccines with some frightening findings. The MfG we found hidden there will also be found throughout the realm of pharmaceuticals, both OTC and Rx ones.
The following excerpt from the recently posted article “What’s in your medicine may surprise you – a call for greater transparency about inactive ingredients,” published by conversation.com will give you an idea of what consumers are up against.
Product labeling for ‘inactive’ ingredients
“As the so-called ‘inactive’ ingredients in medicines, excipients are often mistaken as being free from potential harm. But the evidence suggests otherwise. Between 2015 to 2019, health-care professionals, patients, and manufacturers filed nearly 2,500 reports to the FDA about an adverse reaction to an excipient.
“While excipients are listed on packaging or package insert for over-the-counter and prescription drugs, this information can be difficult to find. Furthermore, patients often switch from brand name to generic versions, or the pharmacist substitutes one manufacturer for another. While the active pharmaceutical ingredient remains the same, excipients may be different, and even seemingly slight differences can significantly impact patient safety. For example, a patient may be allergic to an excipient in the newly refilled medicine with a different manufacturer.
“Excipients are critical materials and serve a broad variety of functions. They serve as fillers, help the body to absorb the medicine, and add flavor or color to drugs. In fact, some are often found in food products, such as lactose, peanut oil, and starch. In the United States, excipients are approved by the FDA as part of the review process for the finished medicine; they are considered by the regulatory agency as generally recognized as safe or ‘GRAS.’ However, a complete picture of their clinical effect remains unclear.
“Research from MIT and Brigham and Women’s Hospital has found that 92.8% of oral medicines contain at least one potential allergen, a concern for individuals with known sensitivities and intolerances. My recent research, investigating the safety of excipients in biologics, which are large complex molecules that are mostly administered through an injection, found case reports of injection site reaction, severe allergic reaction, spike in blood sugar level, and acute kidney failure associated with these ‘inactive’ ingredients.
“Despite some evidence that excipients are responsible for drug reactions, the amount of each excipient added to each drug is not reported for nearly half of biological medicines. In fact, our study found that 44.4% of the biologics’ labels do not list the concentration of the most commonly occurring excipients. This is true for all prescription medicines, not only biologics.
“This lack of information has important implications for patients with diseases prompting dietary restrictions – such as gluten or lactose intolerance, food allergies, or diabetes – because the amount of wheat starch, lactose, peanut oil, and glucose in their medicine can be potentially harmful.”
Knowledge is power. With the FDA working for both Big Food and Big Pharma, knowledge is your greatest asset.
If you have questions or comments, we’d love to hear from you. If you have hints for others on how to avoid exposure to MfG, send them along, too, and we’ll put them up on Facebook. Or you can reach us at questionsaboutmsg@gmail.com and follow us on Twitter @truthlabeling.